2024年9月2日,复宏汉霖(2696.HK)宣布,公司自主开发的帕博利珠单抗生物类似药HLX17(重组抗PD-1人源化单克隆抗体注射液)的临床试验申请获得国家药品监督管理局(NMPA)批准,拟用于治疗黑色素瘤、非小细胞肺癌、食管癌、头颈部鳞状细胞癌、结直肠癌、肝细胞癌、胆道癌、三阴性乳腺癌、微卫星高度不稳定型或错配修复基因缺陷型肿瘤、胃癌等原研帕博利珠单抗在中国获批的所有适应症。

HLX17为复宏汉霖自主开发的帕博利珠单抗生物类似药。按照国家药品监督管理局发布的生物类似药指导原则的要求,并参考欧盟及美国生物类似药指导原则,经药学比对,临床前药理学、药效学、药代动力学和免疫原性研究证明,HLX17与原研帕博利珠单抗相似。
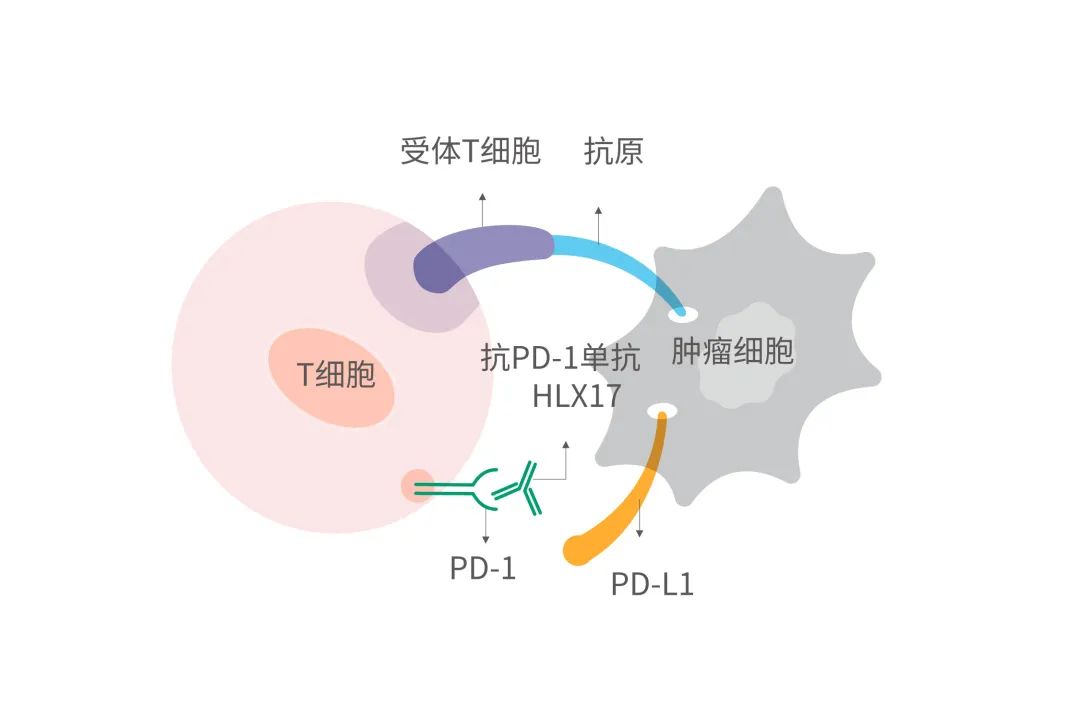
近年来,免疫疗法为肿瘤治疗提供了新的途径,其独特的治疗优势和巨大潜力也陆续得以验证。通过与T细胞上的PD-1受体结合,HLX17能够阻断PD-1与肿瘤细胞上的PD-L1、PD-L2间的相互作用,解除PD-1通路介导的免疫抑制,包括抗肿瘤免疫应答,从而恢复T细胞对肿瘤的免疫监视和杀伤能力,使肿瘤调亡。立足于公司在抗体药物领域的一体化平台优势,复宏汉霖加速免疫治疗药物的开发和布局,围绕PD-1/L1、CTLA-4、LAG-3、TIGIT等免疫检查点已打造出丰富的产品管线,不仅有望在更多适应症中取得突破,也为后续与公司其他产品的协同以及与创新疗法的联合奠定了坚实基础。
未来,复宏汉霖将继续聚焦未满足的临床需求,持续拓宽公司在更多疾病领域的前瞻性布局,为全球患者带去高品质、可负担的创新治疗方案。
关于复宏汉霖
IND Application of Henlius’ Pembrolizumab Biosimilar Approved by NMPA
Shanghai, China, September 2, 2024 - Shanghai Henlius Biotech, Inc. (2696.HK) announced the investigational new drug (IND) application for clinical trial of HLX17, a pembrolizumab biosimilar independently developed by the company, was approved by the National Medical Products Administration (NMPA) for the treatment of indications that reference pembrolizumab has been approved, including melanoma, non-small cell lung cancer, esophageal cancer, head and neck squamous cell cancer, colorectal cancer, hepatocellular carcinoma, biliary tract cancer, triple-negative breast cancer, microsatellite instability-high or mismatch repair deficient cancer, and gastric cancer, etc.
HLX17 is a pembrolizumab biosimilar independently developed by Henlius in accordance with the relevant guiding principles on biosimilar issued by the NMPA, and referencing the guidelines of European Medicines Agency (EMA) and the U.S. Food and Drug Administration (FDA). The pharmacologic comparative study, and preclinical pharmacology study, pharmacodynamics, pharmacokinetics and immunogenicity studies have demonstrated that HLX17 is similar to the reference pembrolizumab.
Immune checkpoint inhibitors are playing a crucial part in immunotherapy, which has emerged in recent years as a novel approach to combating tumor cells and their distinct advantages and enormous potential has been continuously validated. HLX17 is a monoclonal antibody that binds to the PD-1 receptor expressed on T cells and blocks its interaction with PD-L1 and PD-L2, releasing PD-1 pathway-mediated inhibition of the immune response, including the anti-tumor immune response, thus restoring the T-cell immune surveillance of tumors and increasing its anti-tumour efficacy. Based on the competitive edge of an integrated antibody drug R&D platform, Henlius has accelerated the development of immunotherapies and built a diversified product pipeline with high potential immune checkpoints including PD-1/L1, CTLA-4, LAG-3, TIGIT, etc., which are expected to show efficacy in multiple indications while laying a foundation for the synergy with in-house products of the company and other innovative therapies.
Looking forward, Henlius will maintain its focus on unmet medical needs and further broaden the company’s layout in more diseases areas, commit to bring high quality and affordable treatments for patients worldwide.
About Henlius
联系方式
媒体:PR@Henlius.com
投资者:IR@Henlius.com







喜欢本文内容
点击下方按钮·分享 ·收藏 ·点赞 ·在看
























 浙公网安备33011002015279
浙公网安备33011002015279 本网站未发布麻醉药品、精神药品、医疗用毒性药品、放射性药品、戒毒药品和医疗机构制剂的产品信息
本网站未发布麻醉药品、精神药品、医疗用毒性药品、放射性药品、戒毒药品和医疗机构制剂的产品信息
收藏
登录后参与评论